Biomarker Discovery
Biomarkers discovery using protein microarrays involves a multi-step process that combines protein array technology with various experimental and analytical approaches.
A number of good articles have been published for applications like autoimmune diseases, cancer, and many other areas.
Here’s a general overview of the process:
Study design and sample selection: Determine the research question and objectives of your study. Select appropriate sample groups, such as healthy controls and individuals with a specific disease or condition, to compare and identify potential biomarkers.
Protein array selection: Choose the protein array platform that suits your experimental needs. There are different types of protein arrays available, including antibody-based arrays, reverse-phase protein arrays, and peptide arrays. Each array type has its strengths and limitations, so select the one that aligns with your research goals.
Sample preparation: Collect and prepare your biological samples according to standard protocols. Depending on the sample type (e.g., blood, tissue, cell lysates), you may need to perform additional steps such as protein extraction, purification, and quantification.
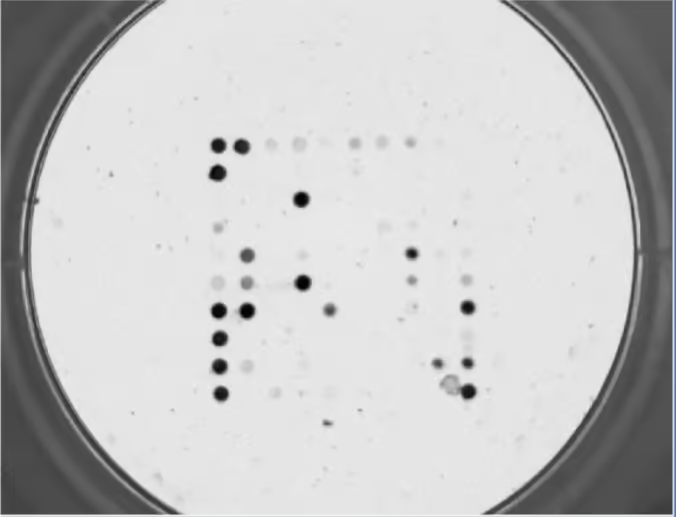
Protein array experimentation: Apply your prepared samples to the protein array platform. The arrays are typically coated with specific capture agents, such as antibodies or peptides, that can selectively bind to target proteins. Incubate your samples with the array, allowing the proteins in the samples to bind to the capture agents on the array surface.
Detection and quantification: After incubation, detect and quantify the proteins bound to the array. This can be achieved using various detection methods, such as fluorescent labeling, chemiluminescence, or mass spectrometry. The specific detection method will depend on the protein array platform you are using.
Data analysis: Analyze the obtained data to identify potential biomarkers. This step involves comparing the protein expression levels between different sample groups (e.g., control vs. disease) and identifying proteins that show significant differences. Statistical analysis methods, such as t-tests, ANOVA, or machine learning algorithms, can be applied to identify statistically significant biomarker candidates.
Validation: Once potential biomarker candidates are identified, further validation is necessary to confirm their association with the studied condition or disease. This can involve using independent sample sets, different experimental techniques (e.g., ELISA), or other validation approaches.
Functional characterization: After validation, it is important to understand the biological relevance and mechanisms of the identified biomarkers. Additional experiments, such as pathway analysis, functional assays, or animal models, can be performed to gain insights into the role of the biomarker in the disease process.
Clinical translation: If the identified biomarkers show promise, they can be further evaluated for their clinical utility and potential for diagnostic, prognostic, or therapeutic applications. This may involve large-scale clinical trials or collaborations with medical professionals to assess their value in patient care.
It’s important to note that the process of biomarkers discovery described above is a general framework, and specific steps may vary depending on the nature of the study, the protein array technology, and the research goals. Additionally, the expertise of researchers in the field is crucial for successful biomarkers discovery using protein arrays..
